Safety Standards for Dental Operatory Illumination
The Critical Role of Illumination in Clinical Excellence and Safety
In any dental practice, the quality of operatory lighting is not a luxury—it is a fundamental component of safe and effective care. Proper illumination directly impacts everything from diagnostic accuracy to the precision of complex restorative procedures. Subpar lighting can lead to misdiagnosed conditions, poor aesthetic outcomes in shade-matching, and significant eye strain for the clinical team, which can degrade performance over a long day. For procurement managers and clinical engineers, understanding the technical standards governing this equipment is paramount. It ensures that every light purchased not only enhances clinical workflow but also meets rigorous international safety and quality benchmarks.
Compliance is the bedrock of trust in all medical devices. For dental lighting, this means adhering to a framework of regulations that guarantee predictable performance, electrical safety, and material biocompatibility. This article provides a detailed guide to the essential safety standards and performance metrics for dental operatory illumination, empowering you to make informed procurement decisions that prioritize patient and practitioner well-being.
More Than Just Brightness: Key Performance Metrics
Experienced clinicians evaluate operatory lights based on measurable performance, not just marketing claims of “brightness.” Focusing solely on lumen output is a common mistake that can lead to poor clinical outcomes. True quality lies in a balanced combination of several key metrics.
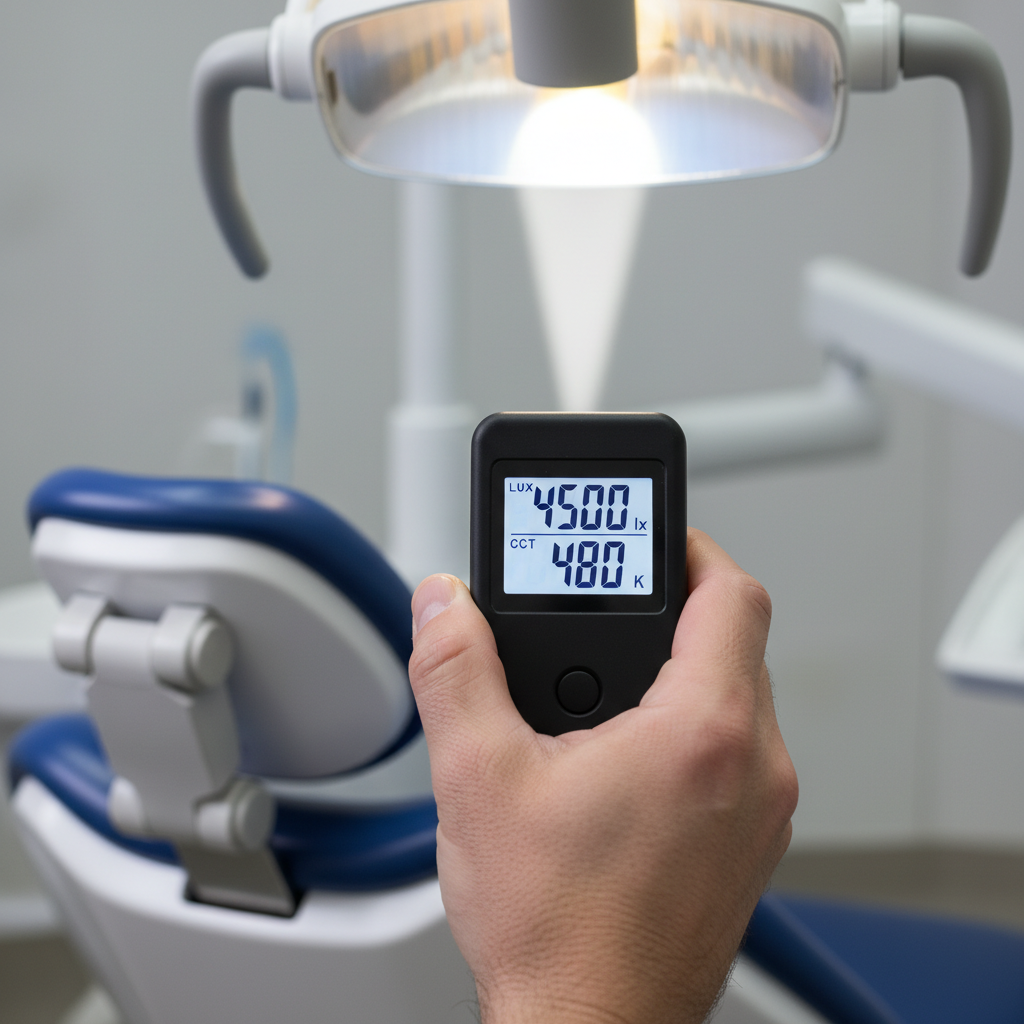
1. Illuminance (Lux) at the Working Surface
Illuminance, measured in lux (lx), defines the amount of light hitting a specific surface area. The required lux level varies depending on the clinical task.
- Examinations: For general oral exams, an illuminance of 10,000–20,000 lux is typically sufficient to identify abnormalities without causing patient discomfort.
- Restorative Procedures: Complex tasks like cavity preparation or crown placement demand higher precision. I aim for a measured illuminance of 30,000–50,000 lux directly on the tooth surface. This ensures clear visibility of fine details and margins.
Pro Tip: Always measure lux at the actual working distance (typically 35–50 cm from the lamp head) with a calibrated light meter. On-site commissioning measurements should be logged in the device’s technical file to satisfy both procurement validation and potential regulatory audits.
2. Color Rendering Index (CRI)
A high CRI is crucial for accurate shade-matching in cosmetic and restorative dentistry. CRI measures how well a light source reveals the true colors of an object compared to a natural light source. A low CRI light can make two slightly different shades appear identical, leading to unsatisfactory aesthetic results.
For any work involving shade-matching, a CRI of 90 or above is the professional standard. I have seen practices invest in high-lux lights with a CRI in the low 80s, only to face persistent issues with crowns and veneers not matching adjacent teeth. This single metric can make or break the aesthetic quality of your work.
3. Color Temperature (Kelvin)
Color temperature, measured in Kelvin (K), describes the appearance of the light, from warm (yellowish) to cool (bluish). For dental applications, a neutral-white light is preferred because it allows for the most accurate perception of tooth and soft tissue colors. The ideal range is generally between 4,000 K and 5,000 K. Light sources outside this range can distort colors, making tissues look inflamed or masking subtle variations in tooth enamel.
4. Beam Quality and Flicker
The geometry of the light beam should provide a focused field of illumination, typically 80–120 mm in diameter at the working distance, to minimize glare for the patient. Furthermore, modern LED lights should be evaluated for flicker. High-frequency flicker, even if imperceptible to the naked eye, can cause headaches and eye strain for the clinical team. Always verify that the LED driver is specified as “flicker-free” or has a flicker percentage below 5%, especially when dimming capabilities are used.
Navigating the Maze of Global Safety Certifications
Ensuring a dental light is compliant means verifying its certifications. These marks are not just logos on a box; they represent a manufacturer’s commitment to quality, safety, and performance as validated by independent or state-sponsored bodies.
European Union: CE Marking and the Medical Device Regulation (MDR)
For any equipment sold within the European Union, the CE mark is mandatory. It signifies that the product meets the high safety, health, and environmental protection requirements of the EU. As explained by the European Commission, the EU Medical Device Regulation (EU MDR 2017/745) has replaced the previous Medical Device Directive (MDD), imposing stricter requirements on manufacturers. This includes more rigorous clinical evaluation, post-market surveillance, and supply chain traceability.
When evaluating a dental light, you must ensure its CE certification is issued under the new MDR standards. This demonstrates a higher level of scrutiny and provides greater assurance of the device’s safety and performance claims. For a deeper understanding of how these regulations apply to the entire operatory, our guide on How Dental Chair Design Meets CE & ISO Safety Standards offers valuable context.
The Global Benchmark: ISO 13485 Quality Management
While the CE mark is specific to the EU, ISO 13485:2016 is the internationally recognized standard for quality management systems in the medical device industry. Certification to this standard demonstrates that a manufacturer has robust processes for the design, production, and distribution of its products.
For a procurement manager, an ISO 13485 certification is a powerful indicator of reliability. It means the manufacturer has systems in place to control product quality, manage risk, and handle post-market feedback. It suggests that the light you buy today will be built to the same standard as the one you buy next year, and that a spare parts plan is likely in place.
United States Market: FDA Regulations
In the United States, medical devices are regulated by the Food and Drug Administration (FDA). Manufacturers must comply with the Quality System Regulation outlined in 21 CFR Part 820, which establishes requirements for current good manufacturing practices (CGMP). These regulations are designed to ensure that medical devices are safe and effective for their intended use. While many international standards harmonize with FDA requirements, compliance with Part 820 is essential for any equipment intended for the U.S. market.
Expert Warning: The Myth of Lumens vs. Lux
A common misconception in purchasing operatory lighting is to confuse lumen output with effective illumination (lux).
- Lumens measure the total amount of light emitted by a source in all directions.
- Lux measures the amount of light that actually falls on a specific surface.
An extremely high-lumen bulb is useless if its optics fail to direct that light precisely onto the operating field. This is why a lower-lumen light with a well-engineered reflector and lens can deliver higher lux at the tooth surface than a high-lumen light with poor beam geometry. Always prioritize measured lux at the working distance and a clean, defined beam pattern over a simple lumen rating.
From Procurement to Practice: A Lifecycle Approach to Lighting
Selecting the right light is only the first step. A long-term strategy for maintenance and validation ensures that performance and safety are sustained throughout the device’s lifespan.
A technician wearing gloves carefully cleans the protective front cover of a dental lamp, demonstrating proper infection control and maintenance.
Pre-Purchase and Commissioning Checklist
To translate these standards into a practical decision framework, I use a checklist to evaluate new lighting systems. This structured approach helps ensure no critical detail is overlooked.
| Metric | Target Specification | Purpose |
|---|---|---|
| Illuminance | 30,000–50,000 lux (Restorative) | Ensures clear visibility of fine details. |
| CRI | ≥ 90 | Guarantees accurate color and shade matching. |
| Color Temperature | 4,000–5,000 K | Provides neutral-white light for true tissue color perception. |
| Beam Diameter | 80–120 mm at working distance | Focuses light on the oral cavity, reducing patient glare. |
| Flicker | < 5% or “Flicker-Free” Driver | Prevents clinician eye strain and camera artifacts. |
| Dimming Range | 10–100% | Allows for adjustment between procedures (e.g., exams vs. composite curing). |
| Certifications | CE (under MDR), ISO 13485 | Verifies compliance with international safety and quality standards. |
Designing for Durability and Maintenance
Long-term reliability depends on thoughtful design and a clear maintenance plan. When inspecting a potential new light, I check for features that simplify upkeep and extend its life.
- Infection Control: The front cover or shield should be removable without tools to allow for thorough cleaning and disinfection. Smooth, non-porous surfaces are essential for preventing cross-contamination. This aligns with principles discussed in our article on Infection Control in Accessible Dental Chair Design.
- Serviceability: A documented 6-to-12-month inspection schedule for checking seals, gaskets, and arm tension is a sign of a well-supported product. Furthermore, the manufacturer should provide a clear 5-year spare parts plan, guaranteeing the availability of critical components like LED drivers, mounts, and control boards. The use of Durable Materials for Long-Term Dental Chair Safety is just as important for lighting as it is for the chair itself.
Key Takeaways
Selecting the right dental operatory illumination goes far beyond choosing the brightest option. It requires a methodical approach grounded in an understanding of international safety standards and practical performance metrics. By prioritizing measured illuminance, high CRI, and appropriate color temperature, you equip your clinical team for success. Verifying certifications like CE (under MDR) and ISO 13485 ensures that you are investing in equipment that is not only effective but also demonstrably safe and reliable. Finally, a proactive maintenance plan protects this investment and guarantees consistent performance for years to come.
Disclaimer: This article is for informational purposes only and does not constitute professional medical or regulatory advice. Always consult with qualified regulatory professionals and clinical engineers to ensure compliance with local and international standards applicable to your practice.






